Benzaldehyde, 2-amino-4-bromo-
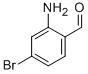
2-Amino-4-bromobenzaldehyde
CAS: 59278-65-8
Molecular Formula: C7H6BrNO
Benzaldehyde, 2-amino-4-bromo- - Names and Identifiers
Benzaldehyde, 2-amino-4-bromo- - Physico-chemical Properties
| Molecular Formula | C7H6BrNO |
| Molar Mass | 200.03 |
| Density | 1.672±0.06 g/cm3(Predicted) |
| Melting Point | 85 °C |
| Boling Point | 317.1±32.0 °C(Predicted) |
| pKa | -0.72±0.10(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Store in freezer, under -20°C |
| Use | Application 2-amino -4-bromobenzaldehyde is an organic synthesis intermediate and pharmaceutical intermediate, which can be used in laboratory research and development process and chemical and pharmaceutical synthesis process. |
Benzaldehyde, 2-amino-4-bromo- - Upstream Downstream Industry
Benzaldehyde, 2-amino-4-bromo- - Reference Information
| overview | 2-amino -4-bromobenzaldehyde can be used as an intermediate in pharmaceutical synthesis, which can be prepared by reaction with 2-amino -4-bromobenzoic acid as the starting material. 2-Amino-4-bromobenzaldehyde can be used to prepare 3-amino -7-bromoquinoline -2-ethyl formate. |
| application | 2-amino -4-bromobenzaldehyde is an organic synthesis intermediate and a pharmaceutical intermediate, which can be used in the laboratory research and development process and the chemical and pharmaceutical synthesis process. |
| preparation | step 1: synthesis of 2-amino -4-bromobenzene methanol 60g NaBH4 is added to 300mL tetrahydrofuran (THF) and stirred mechanically. Under the ice water bath, add 100g of 2-amino-4-bromobenzoic acid to the solution, then add 27mL of boron trifluoride ethyl ether solution dropwise, and control the temperature below 20 degrees. After dropping, raise the reaction solution to room temperature and react overnight until the raw material is finished. Add 300ml MeOH dropwise to the reaction solution, add 5N NaOH(200ml) after the dropwise addition, and stir for about half an hour. The organic solvent in the reaction liquid is spin dried and cooled to room temperature, solid is precipitated, filtered, and the crude 2-amino -4-bromobenzene methanol is obtained by about 60g [mass spectrometry (MS)M 1 203]. 98.7% yield. Step 2: Synthesis of 2-amino-4-bromobenzaldehyde Add 60g and 300g of manganese dioxide obtained in step 1 to 1L dichloromethane solvent, mechanically stir, and react overnight. The reaction liquid is passed through diatomite and column (petroleum ether: ethyl acetate = 5: 1) to obtain about 40g of 2-amino -4-bromobenzaldehyde [nuclear magnetic resonance hydrogen spectrum (400MHz,CDCl3):δ6.72-6.75(d,1h),7.23(bs,2h),7.38-7.42(dd, h),7.72-7.73(d,1h),9.77(s,1H)]. 85.2% yield. |
Last Update:2024-04-09 01:59:58
Supplier List
Spot supply
Product Name: 2-Amino-4-bromobenzaldehyde Visit Supplier Webpage Request for quotationCAS: 59278-65-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple SpecificationsSpot supply
Product Name: 2-Amino-4-bromobenzaldehyde Visit Supplier Webpage Request for quotationCAS: 59278-65-8
Tel: +86-18821248368
Email: Ceya.x@macklin.cn
Mobile: +86-18821248368
QQ: 495145328
Wechat: 17821173903/13918391375
WhatsApp: +86-18821248368
Product Name: 2-AMINO-4-BROMOBENZALDEHYDE Visit Supplier Webpage Request for quotation
CAS: 59278-65-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 59278-65-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Amino-4-bromobenzaldehyde Visit Supplier Webpage Request for quotationCAS: 59278-65-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple SpecificationsSpot supply
Product Name: 2-Amino-4-bromobenzaldehyde Visit Supplier Webpage Request for quotationCAS: 59278-65-8
Tel: +86-18821248368
Email: Ceya.x@macklin.cn
Mobile: +86-18821248368
QQ: 495145328
Wechat: 17821173903/13918391375
WhatsApp: +86-18821248368
Product Name: 2-AMINO-4-BROMOBENZALDEHYDE Visit Supplier Webpage Request for quotation
CAS: 59278-65-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 59278-65-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
Raw Materials for Benzaldehyde, 2-amino-4-bromo-
(E)-2-(4-bromo-2-nitrophenyl)-N,N-dimethylethenamine
4-Bromo-2-nitrobenzoic acid
2-AMINO-4-BROMOBENZYL ALCOHOL
4-Bromo-2-nitrobenzyl alcohol
4-Bromo-2-nitrobenzaldehyde
4-Bromo-2-nitrobenzoic acid
2-AMINO-4-BROMOBENZYL ALCOHOL
4-Bromo-2-nitrobenzyl alcohol
4-Bromo-2-nitrobenzaldehyde
Downstream Products for Benzaldehyde, 2-amino-4-bromo-


