Trihexyl(tetradecyl)phosphonium
TRIHEXYL(TETRADECYL)PHOSPHONIUM TETRAFLUOROBORATE
CAS: 374683-55-3
Molecular Formula: C32H68BF4P
Trihexyl(tetradecyl)phosphonium - Names and Identifiers
Trihexyl(tetradecyl)phosphonium - Physico-chemical Properties
| Molecular Formula | C32H68BF4P |
| Molar Mass | 570.66 |
| Density | 0,93 g/cm3 |
| Melting Point | 38.5 °C |
Trihexyl(tetradecyl)phosphonium - Risk and Safety
| Hazard Symbols | C - Corrosive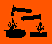 |
| Risk Codes | 34 - Causes burns |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 3261 8/PG 2 |
| WGK Germany | 3 |
| FLUKA BRAND F CODES | 10-21 |
Trihexyl(tetradecyl)phosphonium - References
| Use | trihexyldecatetraalkylphosphorus tetrafluoroborate may be used as the ionic liquid. |
| preparation | ionic liquids containing tetrafluoroborate anions are generally produced as follows: in the second step, the halide, for example, 1-ethyl-3-methylimidazole chloride or bromide, as described in S.Par method, with NaBF4 in acetone; With NaBF4 in water; With AgBF4 or HBF4 in water; With NH4BF4 in acetone; With HBF4 in methanol; or with microwave irradiation and NH4BF4 reaction. However, all known methods have disadvantages, particularly for large-scale industrial synthesis. |
Last Update:2024-04-10 22:29:15
