87-60-5
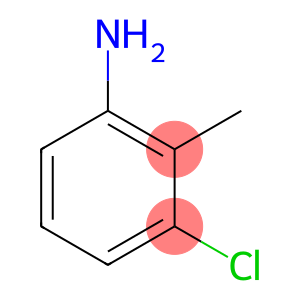
3-Chloro-2-methylaniline
CAS: 87-60-5
Molecular Formula: C7H8ClN
87-60-5 - Names and Identifiers
| Name | 3-Chloro-2-methylaniline |
| Synonyms | 3-Chlor-o-toluidin 3-chloro-2-toluidin 3-chloro-o-toluidin 3-chloro-2-toluidine fast scarlet TR base 3-Chloro-o-toluidine Azoic Diazo Component 46 3-Chloro-2-methylaniline 3-CHLORO-ORTHO-TOLUIDINE 2-Chloro-2-Methyl Aniline 6-Chloro-2-amino-1-methylbenzene |
| CAS | 87-60-5 |
| EINECS | 201-756-6 |
| InChI | InChI=1/C7H8ClN/c1-5-6(8)3-2-4-7(5)9/h2-4H,9H2,1H3 |
| InChIKey | ZUVPLKVDZNDZCM-UHFFFAOYSA-N |
87-60-5 - Physico-chemical Properties
| Molecular Formula | C7H8ClN |
| Molar Mass | 141.6 |
| Density | 1.185 g/mL at 25 °C (lit.) |
| Melting Point | 2 °C (lit.) |
| Boling Point | 115-117 °C/10 mmHg (lit.) |
| Flash Point | >230°F |
| Water Solubility | 1g/L at 20℃ |
| Solubility | 4.5g/l |
| Appearance | Liquid |
| Specific Gravity | 1.19 |
| Color | Clear yellow to red or red-brown |
| BRN | 471697 |
| pKa | pK1:2.49(+1) (25°C) |
| Storage Condition | Store below +30°C. |
| Refractive Index | n20/D 1.588(lit.) |
| Physical and Chemical Properties | Density 1.171 melting point 2-3°C boiling point 115-117°C refractive index 1.588-1.59 flash point 112.8°C |
| Use | Is an important raw material for the synthesis of dyes, pesticides, synthetic dyes DB-50, can produce herbicides such as paddy field |
87-60-5 - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R34 - Causes burns R43 - May cause sensitization by skin contact R51/53 - Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S37/39 - Wear suitable gloves and eye/face protection |
| UN IDs | UN 3429 6.1/PG 3 |
| WGK Germany | 2 |
| RTECS | XU4760000 |
| TSCA | Yes |
| HS Code | 29214300 |
| Hazard Class | 6.1 |
| Packing Group | III |
87-60-5 - Reference Information
| LogP | 2.4 |
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Use | 3-chloro-2-methylaniline is an intermediate of the herbicide quinclorac, which can also be prepared 2,6-dichlorotoluene. The red base TR can be used to dye cotton, viscose fiber, silk, nylon and acetate fiber, and can also be used for cotton printing. Mainly used for dyeing red, such AS with the phenol AS, AS-BO, AS-ITR and other coupling are red. The coupling ability is medium, and the coupling speed is fast. intermediates in organic synthesis. is an important raw material for the synthesis of dyes, pesticides, can be synthesized dye DB-50, can be produced in paddy field herbicide fast kill, etc. |
| production method | O-nitrotoluene is obtained by chlorination and reduction. 1. Chlorination in O-nitrotoluene, add 2% ferric chloride as a catalyst, at 50-60 deg C into chlorine, the relative density of the reaction solution reaches 1.27-1.29(50 Deg C), it was washed with 5% hydrochloric acid, washed with water, and finally adjusted to pH 7 with liquid alkali. The water layer was separated and distilled off, followed by distillation under reduced pressure to collect the 120-150 ° C. (5.33-8.0kPa) fraction, which was cooled to crystallize and filtered to obtain 2-chloro-6-nitrotoluene. 2. Reduction hydrochloric acid and iron powder were added to the reaction Pan, heated with stirring to 90 ° C., and melted 2-chloro-6-nitrotoluene was slowly added, and the mixture was refluxed for 3H. Cooling, adding liquid alkali to adjust the pH to 8, and steam distillation to obtain 3-chloro-o-toluidine. products were obtained from O-nitrotoluene by chlorination and reduction. The product was purified by distillation and then salified with hydrochloric acid, filtered, dried and pulverized to obtain a finished product.. The preparation method is to put 2-chloro-6-nitrotoluene, Catalyst Raney nickel, auxiliary agent and methanol in a pressure kettle, and react with hydrogen for 2H at a certain temperature to remove the solvent, 3-chloro-2-methylaniline was obtained by distillation under reduced pressure. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 02:00:12
Supplier List
Featured ProductsMultiple SpecificationsSpot supply
Product Name: 3-Chloro-2-methylaniline Visit Supplier Webpage Request for quotationCAS: 87-60-5
Tel: +86-18821248368
Email: Ceya.x@macklin.cn
Mobile: +86-18821248368
QQ: 495145328
Wechat: 17821173903/13918391375
WhatsApp: +86-18821248368
Spot supply
Product Name: 3-Chloro-2-methylaniline Request for quotationCAS: 87-60-5
Tel: +86-17551318830
Email: r@reformchem.com
Mobile: +86-17551318830
QQ: 3787852685
Multiple Specifications
Product Name: 3-Chloro-2-methylaniline Request for quotationCAS: 87-60-5
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Spot supply
Product Name: 3-Chloro-2-methylaniline Request for quotationCAS: 87-60-5
Tel: +86-17551318830
Email: r@reformchem.com
Mobile: +86-17551318830
QQ: 3785839865
Spot supply
Product Name: 3-Chloro-2-methylaniline Request for quotationCAS: 87-60-5
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
Spot supply
Product Name: 3-Chloro-2-methylaniline Visit Supplier Webpage Request for quotationCAS: 87-60-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Chloro-2-methylaniline Request for quotation
CAS: 87-60-5
Tel: +86-571-88162785
Email: Lucy@verypharm.com
Mobile: +86-13606544505
QQ: 472954302
Product List: View Catalog
CAS: 87-60-5
Tel: +86-571-88162785
Email: Lucy@verypharm.com
Mobile: +86-13606544505
QQ: 472954302
Product List: View Catalog
Featured ProductsMultiple SpecificationsSpot supply
Product Name: 3-Chloro-2-methylaniline Visit Supplier Webpage Request for quotationCAS: 87-60-5
Tel: +86-18821248368
Email: Ceya.x@macklin.cn
Mobile: +86-18821248368
QQ: 495145328
Wechat: 17821173903/13918391375
WhatsApp: +86-18821248368
Spot supply
Product Name: 3-Chloro-2-methylaniline Request for quotationCAS: 87-60-5
Tel: +86-17551318830
Email: r@reformchem.com
Mobile: +86-17551318830
QQ: 3787852685
Multiple Specifications
Product Name: 3-Chloro-2-methylaniline Request for quotationCAS: 87-60-5
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Spot supply
Product Name: 3-Chloro-2-methylaniline Request for quotationCAS: 87-60-5
Tel: +86-17551318830
Email: r@reformchem.com
Mobile: +86-17551318830
QQ: 3785839865
Spot supply
Product Name: 3-Chloro-2-methylaniline Request for quotationCAS: 87-60-5
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
Spot supply
Product Name: 3-Chloro-2-methylaniline Visit Supplier Webpage Request for quotationCAS: 87-60-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Chloro-2-methylaniline Request for quotation
CAS: 87-60-5
Tel: +86-571-88162785
Email: Lucy@verypharm.com
Mobile: +86-13606544505
QQ: 472954302
Product List: View Catalog
CAS: 87-60-5
Tel: +86-571-88162785
Email: Lucy@verypharm.com
Mobile: +86-13606544505
QQ: 472954302
Product List: View Catalog
View History






