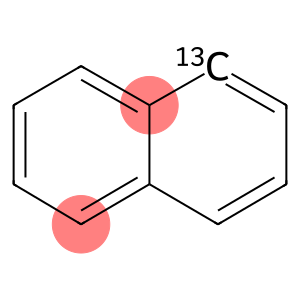
NAPHTHALENE-1-13C
Naphthalene-1-13C
CAS: 20526-83-4
Molecular Formula: C10H8
NAPHTHALENE-1-13C - Names and Identifiers
| Name | Naphthalene-1-13C |
| Synonyms | NAPHTHALENE-1-13C Naphthalene-1-13C |
| CAS | 20526-83-4 |
NAPHTHALENE-1-13C - Physico-chemical Properties
| Molecular Formula | C10H8 |
| Molar Mass | 129.18 |
| Melting Point | 80-82°C(lit.) |
| Boling Point | 218°C(lit.) |
| Flash Point | 80℃ |
| Storage Condition | Room Temprature |
NAPHTHALENE-1-13C - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R40 - Limited evidence of a carcinogenic effect R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S36/37 - Wear suitable protective clothing and gloves. S46 - If swallowed, seek medical advice immediately and show this container or label. S60 - This material and its container must be disposed of as hazardous waste. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN 1334 4.1/PG 3 |
| WGK Germany | 3 |
NAPHTHALENE-1-13C - Introduction
Naphthalene-1-13C is a naphthalene compound with the isotope carbon -13 substituted for the carbon atom. Its chemical structure is similar to ordinary naphthalene, but one of the carbon atoms is replaced by carbon -13.
Naphthalene-1-13C has many properties similar to those of ordinary naphthalene. It is a solid with a strong aromatic smell. It has a melting point of 79-82°C and a boiling point of 216-218°C. It is soluble in organic solvents such as benzene and methylene chloride at normal temperature.
Naphthalene-1-13C are widely used in scientific research. The isotopic labeling of carbon -13 allows the compound to be used to study molecular structures and reactions. By monitoring the carbon -13 nuclear magnetic resonance spectrum, the chemical environment and relative positions of different carbon atoms in the naphthalene molecule can be determined. Therefore, Naphthalene-1-13C are often used in pharmaceutical research, chemical reaction kinetics and nuclear magnetic resonance spectroscopy.
The method of preparing Naphthalene-1-13C is mainly realized by organic synthesis chemical reaction. A common preparation method is through the reaction of a selective alkyl lithium reagent with 13C-labeled iodonaphthalene, followed by mass spectrometric verification of carbon -13 stable isotope labeling. The specific preparation details can be adjusted according to different laboratories and needs.
For Naphthalene-1-13C safety information, because its chemical properties are similar to ordinary naphthalene, general safety measures apply. Inhalation, ingestion and skin contact should be avoided. Wear protective glasses, gloves and laboratory coats when using. Follow appropriate laboratory practices and waste disposal regulations during use and disposal. In the event of an accidental leak, appropriate measures should be taken immediately to clean up and dispose of it.
Naphthalene-1-13C has many properties similar to those of ordinary naphthalene. It is a solid with a strong aromatic smell. It has a melting point of 79-82°C and a boiling point of 216-218°C. It is soluble in organic solvents such as benzene and methylene chloride at normal temperature.
Naphthalene-1-13C are widely used in scientific research. The isotopic labeling of carbon -13 allows the compound to be used to study molecular structures and reactions. By monitoring the carbon -13 nuclear magnetic resonance spectrum, the chemical environment and relative positions of different carbon atoms in the naphthalene molecule can be determined. Therefore, Naphthalene-1-13C are often used in pharmaceutical research, chemical reaction kinetics and nuclear magnetic resonance spectroscopy.
The method of preparing Naphthalene-1-13C is mainly realized by organic synthesis chemical reaction. A common preparation method is through the reaction of a selective alkyl lithium reagent with 13C-labeled iodonaphthalene, followed by mass spectrometric verification of carbon -13 stable isotope labeling. The specific preparation details can be adjusted according to different laboratories and needs.
For Naphthalene-1-13C safety information, because its chemical properties are similar to ordinary naphthalene, general safety measures apply. Inhalation, ingestion and skin contact should be avoided. Wear protective glasses, gloves and laboratory coats when using. Follow appropriate laboratory practices and waste disposal regulations during use and disposal. In the event of an accidental leak, appropriate measures should be taken immediately to clean up and dispose of it.
Last Update:2024-04-09 21:04:16
Supplier List
Product Name: Naphthalene-1-13C Visit Supplier Webpage Request for quotation
CAS: 20526-83-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 20526-83-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Naphthalene-1-13C Visit Supplier Webpage Request for quotation
CAS: 20526-83-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 20526-83-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
